The New Armenian medical journal
Vol.12 (2018), Nо3, p. 49-57
Endoscopy in early gastric cancer diagnosis: step by step (surgical perspective)
Levkin V.V., Kharnas S.S., Mirijanyan M.M., Shestakov A.M., Lesko K.A., Kulkova R.A.
1 Department of Surgery No 1, I.M. Sechenov First Moscow State Medical University, Moscow, Russia
2 Department of Operative Surgery and Topographic Anatomy, I.M. Sechenov First Moscow State Medical University, Moscow, Russia
3 Faculty of General Medicine, Yerevan State Medical University after M. Heratsi, Yerevan, Armenia
4 Department of Radiology, Loginov Moscow Clinical Scientific Center, Moscow, Russia
5 Department of the Russian Language and Literature, Sang-Myung University, Cheonan Sity, Korea
ABSTRACT
Gastric cancer is one of the leading causes of oncologic morbidity and mortality both in Russia and worldwide. The primary method for early diagnosis of gastric cancer and upper gastrointestinal tract is endoscopy. Present article discusses the evolution of this method. The authors demonstrate the picture of the method’s development from first devices introduced in the early 19th century to the period of the endoscopic technology flourishing in the late 20th century resulting in the increasing rate of early gastric cancer detection. They pay a special attention to the innovative endoscopic technologies analyzing the advantages and drawbacks. The article covers both traditional tests including ultrasound test and narrowband imaging and new methods bringing endoscopy to the new level of visualization. The latter involve confocal laser endomicroscopy and endocytoscopy. The development of medicine in general, and oncology in particular, over the past decades tends to fetishize laboratory and instrumental techniques over the knowledge of the clinical symptoms of various diseases. In the diagnosis of stomach diseases, the clinical manifestation as well as knowledge of risk groups is of crucial importance for the selection of patients for special tests, and primarily for endoscopy. This approach enables the identification of patients at an early stage of the disease and will not only improve long-term treatment results, reduce mortality from gastric cancer, but it will improve the quality of operated patients’ life through the active introduction of intraluminal manipulations (endoscopic resection of the mucosa with dissection in the submucosal layer and others) in gastric cancer which are much less invasive compared to standard subtotal stomach resection or gastrectomy with enlarged lymphadenectomy (D2), still widely performed in all cases of resectable cancer irrespective of its stage (I, II or III). Summing up the upper gastrointestinal tract endoscopic diagnosis development, the authors emphasize the importance of the introduction of modern endoscopic diagnosis and treatment as well as an individualized clinical approach to the patient examination and the adequate treatment selection.
Introduction
Among gastrointestinal tract diseases, gastric cancer is especially significant since it is one of the most common oncological conditions worldwide with around 1 million gastric cancer cases registered annually and with over 800 thousand people dying within the first year after diagnosis. In the Russian Federation, as in most countries of the world, gastric cancer is diagnosed in almost 50% of the patients at stages III-IV, and 5-year survival rate is generally 25-30%. with gastric cancer diagnosed at the early stages, 5-year survival rate may be over 90%. A favorable prognosis is based on low rate of relapse, and lymphogenous and hematogenous metastasis [Ryabov A et al., 2015]. Unfortunately, the proportion of early diagnosis of gastric cancer in most countries of the world, including the Russian Federation, is rarely over 10% [Kuvaev R et al., 2014]. However, Japan and South Korea (countries with the highest gastric cancer prevalence), the early diagnosis rate of gastric cancer is 50% and even higher [Bray F et al., 2013]. Thus, the development of early diagnosis methods is not only reasonable, but also vitally important. Currently, the most informative early diagnosis of gastric cancer is gastroscopy based on modern specifying technologies. However, prior these technologies were developed, endoscopy passed a long, more than 200-year evolution. For many centuries, physicians dreamed about the visual evaluation of the upper gastrointestinal tract, not to mention the early diagnosis of gastric cancer, with various optical devices [Balalykin D, 2008; 2009 a, b, c]. what was the problem? The technical difficulties were combined with topographic and anatomical features upper gastrointestinal tract and pharynx (pharynx). The instrument insertion through the oropharynx (pars oralis pharynges) and the laryngeal pharynges (pars laryngea pharynges) required certain caution. One should also keep in mind the of lymphoid tissue accumulation, i.e. the tonsil pharyngealis at the transition of the upper pharyngeal wall to the middle one; the pharyngeal muscles, which include three pharyngeal constrictors (m | m constrictor pharyngis superior, middle and inferior), and pharyngeal lifters: the tubular-pharyngeal muscle (m.salpingopharyngeus) and the stilopharyngeus muscle (m. stilopharyngeus). The transition of the lower pharynx to the esophagus forms an angle at the level of the sixth cervical vertebra requiring either a flexible instrument or an extremely “non-physiological position” of a patient. Two esophageal physiological narrowings (at the level of the left main bronchus crossing and passing through the esophagus), as well as the right and left esophageal deviation did not pose problems due to good elasticity of the esophagus wall, which consists of mucosal (tunica mucosa) and submucosal (tunica submucosa) membranes, muscle tunic (tunica muscularis) and serous membrane (tunica serosa) [Wood W et al., 2010].
The stomach sections that were not available for visual inspection with a rigid instrument – the fundus ventriculi and the canalis pyloricum, and the duodenum – made the stomach examination difficult if possible at all. Nevertheless, the technological progress helped to overcome difficulties associated with the anatomic features of the upper gastrointestinal tract. The modern techniques can reveal both early gastric cancer and mucosal precancerous conditions. what was the evolution the endoscopic devices? the endoscopy development: In the early 19th century, Frankfurt-am-Main physician Ph. Bozzini put forward the concept of visual examination of the upper gastrointestinal tract [Balalykin D, 2008; 2009 a, b, c]. His device enabled indirect esophagoscopy of the upper esophagus only. For a long time, those devices were rigid and often less than 10cm long. The light source was a candle later replaced with a kerosene lamp. In the 1880s, optical prisms, electric bulbs and bending tubes were introduced enabling the first successful endoscopic examination of the entire esophagus and the stomach. I. Mikulicz suggested direct rather than reflected vision in the endoscopy thus laying the foundation of gastroscopy [Balalykin D, 2008; 2009 a, b, c]. The imperfection of the endoscopic devices made these manipulations difficult to tolerate, dangerous and time consuming. Nevertheless, hundreds of successful esophageal examinations were performed. Moreover, the technique was used not only for diagnosis but also for the foreign body removal from the esophagus (V. Hacker). In the late 19th century, Russian physician P.M.Revidov designed a flexible endoscope: a system of three tubes – the external flexible tube and two internal tubes inserted into one another [Balalykin D, 2008; 2009 a, b, c]. The internal tube was inserted as a rubber stomach probe, the other two tubes contained optical system and an electric bulb for illumination. In the early 20th century, the first endoscopic guidelines and atlases with watercolor illustrations were published. The number of successful examinations gradually grew up however the technique was not clinically significant. R. Schindler who designed semiflexible lens endoscope made the important contribution into the endoscopy development thus laying the foundation of modern gastroscopy (1932).
Schindler’s gastroscope included a proximal metal section and a flexible distal section with a prism and an electric bulb. A Richardson cylinder and an ocular with an illumination system were mounted on the proximal section. with small changes, this endoscope was produced in many countries including the USSR until the late 1950s. However, only a limited number of physicians performed endoscopic esophageal and gastric examinations (duodenal examination was still unavailable) in several large clinics. Until the mid-1960s, the widely available diagnostic method for upper gastrointestinal tract diseases – especially for gastric cancer diagnosis – was x-ray test. The indications for endoscopic examination were a suspected gastric cancer or inability to exclude gastric cancer [Balalykin D, 2008; 2009 a, b, c]. A breakthrough in endoscopy was the development of a flexible optic fiber endoscope in 1958 by the group of physicians led by B.I. Hirschowitz who used the invention of van Heel. The new fiberscope was flexible and outmatched the light and color characteristics of previous devices. Besides a thorough examination of the gastric bottom and cardia (in retroflexio), which was problematic for the previous generation of endoscopes, the fibroscope enabled duodenoscopy. The serial production of the fiberscope started at Japanese companies (Olympus and others) in the early 1960s. Fiber-optic technology allowed the diagnosis of the upper gastrointestinal tract benign and malignant diseases and a targeted biopsy of the mucous membrane. This was a targeted biopsy, semi-flexible endoscopes enabled a biopsy but it was less accurate. Moreover, new manipulations were introduced into the clinical practice: polypectomy, gastric mucosa endoscopic resection in early cancer patients, interventions on the large duodenal papilla, contrasting and stenting of the biliary and pancreatic ducts, etc. However, the fibergastroscopy is not without flaws. There are certain difficulties in identifying early forms of neoplasias, areas of dysplasia and metaplasia, common forms of cancer with a submucous growth, differentiation of the stomach benign and malignant diseases (especially peptic ulcer and infiltrative-ulcerative form of cancer). Nevertheless, the introduction of fibergastroscopy improved the endoscopy quality and increased the rate early gastric cancer detection from several percent to 30%.
The success of surgical treatment depends on the timely diagnosis of gastric cancer. Therefore, the further developments in endoscopy were primarily aimed at precancerous conditions detection and improvement of early gastric cancer diagnosis. Initially, a method of chromoendoscopy was developed: the intravital staining of the organ mucous membrane to identify changes not revealed in a routine endoscopic examination and to perform a targeted biopsy. Various dyes were used: methylene blue, indigo carmine, etc. Chromoendoscopy improved the diagnosis quality, especially of early gastric cancer. Chromoendoscopy shortcomings were: 1) labor intensity, 2) duration of the procedure, 3). staining of the erosive and ulcerative lesions of the neoplastic genesis, 4) low effectiveness in a submucosal tumor, 5) no dye tropic to tumor tissue. Therefore, chromoendoscopy application was limited [Poddubny B et al., 2005, Sokolov V, 2015]. In the 1990s, high quality and high-resolution video endoscopes with computer image analysis were developed and introduced into clinical practice [Poddubny B et al., 2005, Sokolov V, 2015]. However, fiber-optic technology still remained a method of choice in some settings. The equipment was considered relatively cheap, subject to various repair improvements, although it had a limited lifespan due to the fiber fragility. The drawbacks include the inability to improve the image without the increase of light fiber number, which required the device diameter enlargement up to 12-14 mm. Moreover, the endoscope should be kept at eye level all the time, which is inconvenient. At present, fiberscopes are used primarily in urgent surgery, and for intraoperative endoscopy. Most leading companies continue the serial production of fiberscopes though in less quantities than video endoscopes. As for the benefits of video endoscopy, it primarily involves the opportunity for several specialists to simultaneously view on the monitor, to enlarge and save the image [Puddubny B et al., 2005, Sokolov V, 2015]. Further improvement of video endoscopes involved image enlargement and its resolution increase. Now there are two methods of magnifying (zoom) endoscopy – electronic (digital) based on the image processing and optical based on a system of coaxial magnifying lenses. The methods enable optical and digital picture enlargement by five and 80-100 times, respectively. About 10 years ago, high resolution video endoscopy systems were developed with increased pixels per square inch parameter. Image quality is comparable to that of magnifying endoscopy [Sokolov V, 2015]. endoscopic diagnosis prospects: Unfortunately, neither magnifying video endoscopy nor high-definition endoscopy can reveal structural changes in the mucosa, and in particular, the micro vascular structural disorders characteristic to tumor transformation even at the earliest stage of the gastric cancer, which cannot be detected visually. what does it happen? The normal gastric mucosa looks pink in any endoscopic technique (fiber-optic or video), as it absorbs light in the blue and green parts of the spectrum and reflects mostly in the red part of the spectrum. This results from the application of xenon light sources that emit light in wavelengths from 400 (blue) to 700 nm (near infrared) [Gono K et al., 2003; 2004]. If we use not the entire spectrum but some of the bands or a combination of them to illuminate the mucosa, the picture will look quite different, which makes it possible to detect changes that are not identifiable in white light [Gono K et al., 2003; Sokolov V, 2015]. This is the basis of such refining endoscopic technologies as narrow-band imaging (NBI), flexible spectral imaging color enhancement (FICE), and infrared laser (> 700 nm) video endoscopy. Narrow-band imaging is a widely used technique available in all modern endoscopic units. The NBI procedure is as follows: after a visual evaluation in white light, the mucosa is illuminated with two narrow spectrum bands – blue (390-460 nm) and green (530-560 nm), which are the absorption bands of hemoglobin. The isolation of these bands involves a system of special light filter. Blue light diffuses in the mucosal surface layers enabling capillary visualization and revealing the mucous membrane surface relief. Green light penetrates deeper into the tissue and revealing the venules in the submucosal layer. On the monitor, the surface of the unchanged mucosa looks light green, the capillaries look brown, the venules look light blue [Gono K et al., 2003; Sokolov V, 2015]. The comparative clinical studies indicate that the accuracy of narrow-spectrum imaging in identifying pre-cancer and early gastric cancer foci is comparable to that of chromoendovideoscopy, but requires less time and effort. The combination of magnifying endoscopy and NBI
enables to identify with high probability partial gastric mucosal malignant transformation based on vascular changes: dilatation, tortuosity, caliber changes and different vascular shape and some other signs [Kuvaev R et al., 2014]. More recently, the FICE technique was developed, based on physical principles other than NBI [Osawa H, Yamamoto H, 2014]. Flexible spectral imaging color enhancement involves white light from the xenon lamp (unlike the blue-green light in NBI) sent through the endoscope to the area of interest. The endoscope CCD-matrix records the reflected light and converts it into an endoscopic image on the monitor. The FICE limits spectral range of the reflected light signal and displays images in specific wavelengths. For example, the recommended spectral band combination for the gastric mucosa examination is: 520 nm (red), 500 (green) and 405 (blue). The method looks promising [Sokolov V, 2015]. Infrared laser (> 700 nm) video endoscopy involves preliminary infusion of indocyanine green as a fluorophore contrasting the small vessels and enabling targeted examination of vessels in the submucosal layer, identification of interstitial tissue microvascular structures characteristic to a tumor and specification of the tumor lesion depth in submucosal tumor infiltration [Sokolov V, 2015]. In recent years, video endoscopes with the virtual spectroscopy function for the hemoglobin index were developed. This function enables the vascular structure analysis based on the hemoglobin concentration in the mucous membrane areas of interest. The test produces a color image, similar to the thyroid gland scintigraphy image for visual assessment. The method is at the clinical trial stage [Sokolov V, 2015]. One of the promising methods of early diagnosis of malignant tumors, including gastric cancer, is laser induced fluorescence spectroscopy – “optical biopsy”. The method involves autofluorescence, which is primarily fluorescence of endogenous porphyrins, whose concentration in tumor cells is higher than in normal cells, and secondary fluorescence of special exogenous photosensitizers tropic to tumor cells. Various photosensitizers were developed with different chemical structure (phtalal derivatives, porphyrins, chlorines, etc.). Fluorescence induction results from laser irradiation with wavelength from 300-500 to 700-1200 nm. The 600-1200 nm (red-infrared radiation) band provides optimal tissue penetration of up to 3 mm. These penetration depth and fluorescence spectra registrations provide reliable information for submucosal gastric tumor diagnosis [Loshchenov V et al., 2013]. None of methods is flawless. Thus, autofluorescence requires highly sensitive and therefore expensive equipment. In secondary fluorescence, photosensitizers remain not only in the tumor, but also in the skin and visible mucous membranes for several days to several weeks. This requires skin protection to prevent photodermatitis. Laser induces fluorescence spectroscopy got a new impetus following the synthesis of 5-aminolevulinic acid [Loshchenov V et al., 2013]. The substance is produced in the human body (heme precursor). The substance is not a photosensitizer. However, it induces photoactive protoporphyrin Ix synthesis and accumulation in tumor cells resulting in more intensive malignant tumor fluorescence relative to the adjacent tissues, thus providing a more reliable diagnosis compared to autofluorescence. Moreover, the intensity of 5-aminolevulinic acid-induced fluorescence is so high that it can be detected in the tumor not only with a spectral analyzer, but also with a special highly sensitive camera, which looks very promising for clinical practice. Currently, prototype endoscopes, including a video fluorescent gastroscope based on aminolevulinic acid-induced fluorescence are under development. It is noteworthy that a serial endoscopic system capable of autofluorescence registration was developed based on a different principle. The 500 +/- 20 nm irradiation (and not laser irradiation as in the system described above) induces the glowing of natural submucosal layer fluorophores, which is recorded by endoscope matrix without significant absorption in the mucosa. The mucosal thickening resulting from, for example, the tumor transformation weakens the signal and purplish-violet areas appear on the green background of the unchanged mucosa. However, the sensitivity and specificity of the method is still low [Kuvaev R et al., 2014]. In the 1990s, endoscopic and ultrasound tests were combined in a single device resulting in the development of endoscopic ultrasonography [Poddubny B et al., 2005, Sokolov V, 2015]. Endoultra sound is the most accurate preoperative method to determine the tumor invasion depth (T) in gastric cancer with the test efficacy of 92%. The test provides the greatest accuracy in the evaluation of a tumor in the mucosal-submucosal layer, and endoultrasound is used to select patients for endoscopic mucosal resection in early gastric cancer. Modern instruments allow visualization of 1-2 mm structures and detection of 5-7 gastric wall layers depending on the sensor frequency characteristics. One of the signs of tumor lesion is a disorder or complete destruction of the stomach wall layered structure. The test is performed with a special echo endoscope with an ultrasound transducer (convection, linear or sector), or an ultrasound probe inserted through the instrumental channel of a standard endoscope. Moreover, linear echo endoscopes enable a targeted biopsy of the stomach wall and the perigastric lymph nodes, and ascitic fluid sampling [Caletti G, Fusaroli P, 1993; Caletti G et al., 2001]. The stenosis of the stomach cardia prevents the endoscope insertion into the stomach making an endoultrasound test impossible; the diagnostic value of the method is also significantly reduced in case of the gastric antral stenosis. Another direction of endoscopy development is an intravital morphological evaluation of the existing changes in the mucosa: endocytoscopy, confocal laser microscopy and optical coherence tomography [Kitabatake S et al., 2006; Testoni P, Mangiavillano B, 2008; Sokolov V, 2015]. what are physical basis of each method? Endocytoscopy is a method of vital endoscopic cytological research. As with a conventional cytological examination under a microscope, a direct magnification of the image (up to 1350 times) occurs after the mucosa pre-staining with a special dye to visualize the cell nuclei. Based on the polymorphism of the cell nuclei, one can evaluate the possible tumor transformation in the mucosa. The stomach examination involves 1% solution of the toluidine blue dye. After the necessary exposure, the dye surplus is thoroughly washed off; the study is performed by direct contact of the endocytoscope with the site under study. There are two methods of research (as in endoultrasound): with the special probes inserted into the biopsy channel of the video endoscope, and with endocytoscopic devices mounted in the video endoscope. The max imum diagnostic accuracy requires combined evaluation by an endoscopist and a morphologist. Optical coherence tomography is an optical investigation method, which provides high-resolution cross-sectional images of the body tissues and enables microscopic morphological assessment. The equipment involves infrared light, rather than ultrasonic acoustic waves as with endoscopic-ultrasound test [Testoni P, Mangiavillano B, 2008]. The optical beam focuses on the tissue, and the echo-delay of the light reflected from the internal microstructure at different depths is measured with an interferometer. A microprobe combined with standard endoscopes enables optical coherence tomography of the gastrointestinal tract mucous membrane and submucosal. Compared to endoultrasound test, optical coherence tomography has an order of magnitude greater resolution and an order of magnitude less scanning depth (up to 2 mm). The best images were obtained from the esophagus (with Barrett’s esophagus) and the colon. Another method that enables the assessment of the mucosal morphological structure in vivo and in real time is the confocal laser endomicroscopy [Kitabatake S., et al, 2006]. There are several types of it, but only fluorescent confocal laser endomicroscopy is suitable for the gastrointestinal tract. The test involves infusion of 10% fluorescein solution used a photosensitizer several minutes prior the test. The drug is well tolerated, but allergic reactions and even short-term jaundice are possible. The laser wavelength for fluorescence induction is usually 488 nm, the response fluorescence is recorded in the 505-585 nm band. The area of the mucosal surface being examined is about 600 μm, and the scanning depth is up to 250 μm (an order of magnitude smaller than with endoscopic optical coherence tomography). The study is performed either with a special confocal endoscope or with a probe with a similar device, which is inserted through the endoscope biopsy channel. The distal section of the endoscope or probe is positioned perpendicular to the contacting surface. The area for scanning is selected after a preliminary visual evaluation with conventional video endoscopy. This technique provides a recordable dynamic monochrome microscopic image of the mucosa structure. This picture is more informative for a morphologist (as with the endocytoscopy) rather than to an endoscopist. The confocal laser endomicroscopy disadvantage is the impossibility of a direct visual evaluation of cell nuclei, such as for endocytoscopy, and lack of a safe drug to stain cell nuclei. The method is at the clinical development stage. Over two centuries passed from the development of the very idea to examine the intestinal tract from the inside. The progress in visual examination of the upper gastrointestinal tract was slow and difficult. Initially, it was indirect (reflected) endoscopy with a rigid endoscope. The examination was limited to the upper esophagus. This method was replaced with direct endoscopy with semi-rigid endoscopes, which enabled the examination of the entire esophagus and even the stomach. After decades, the era of serial semi-rigid endoscopes, able to examine the entire stomach, began. Only in the 1960s and 1970s, the flexible fiber-optic gastroendoscope was developed and put into serial production. Fiber-optic technology enabled the examination of the duodenum. Today, endoscopy is not limited to visual examination of the mucous membranes of the organs of the upper gastrointestinal tract. Many modern techniques accumulate several technological solutions (for example, visual examination and intraluminal ultrasound test, or examination and microscopy). They are now introduced into the national and international clinical practice. Magnifying video endoscopy, high quality endoscopy, endoultrasound and NBI are in the arsenal of modern endoscopic units. However, if magnifying endoscopy and high quality endoscopy are routinely used in many medical institutions, the endoultrasound and NBI are refining diagnosis methods. The indications for NBI (and FICE in the future) are eroded mucosa, superficial ulceration, areas with uneven tuberosity, as well as scars and polypoid formations. Other techniques are in the stages of clinical trials or clinical experience accumulation. After a while we may see new technological solutions and breakthroughs in this area, resulting primarily from the combination of several methods (for example, video fluorescence esophagogastroduodenoscopy with 5-aminolevulinic acid). The following pictures demonstrate early cancer diagnostic capabilities as well as gastric mucosa detalization in the images produced with modern endoscopic technologies (Fig. 1-5).
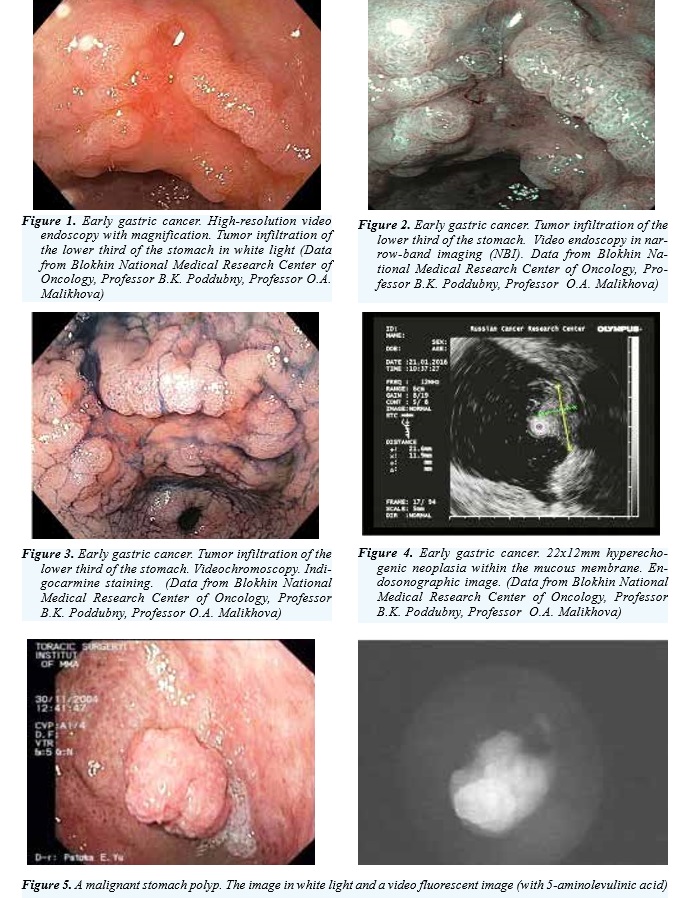
Conclusion
The development of medicine in general and oncology in particular over the past decades tends to fetishize laboratory and instrumental techniques over the knowledge of the clinical symptoms of various diseases. In the diagnosis of stomach diseases, the clinical manifestation as well as knowledge of risk groups is of crucial importance for the selection of patients for special tests, and primarily for endoscopy [Kaprin A, 2015]. This approach enables the identification of patients at an early stage of the disease and will not only improve long-term treatment results, reduce mortality from gastric cancer, but it will improve the quality of operated patients’ life through the active introduction of intraluminal manipulations (endoscopic resection of the mucosa with dissection in the submucosal layer and others) in gastric cancer which are much less invasive compared to standard subtotal stomach resection or gastrectomy with enlarged lymphadenectomy (D2), still widely performed in all cases of resectable cancer irrespective of its stage (I, II or III) [Ryabov A et al., 2015].
REFERENCES
1. Balalykin DA. [An endoscopic exam in the middle of xx century. Part I] [Published in Russian]. Bulletin of surgical gastroenterology. 2009a; 3: 74-79.
2. Balalykin DA. [An origin of endoscopic methods of diagnosis and treatment of esophageal and stomach diseases in early ХIХ-ХХ centuries. Part 1 and 2] [Published in Russian]. Bulletin of surgical gastroenterology. 2008; 2: 74-86.
3. Balalykin DA. [An origin of esophagogastroscopy in ХIХ-ХХ centuries]. [Published in Russian]. Surgery. 2009b; 2: 86-88.
4. Balalykin DA. [Endoscopy of stomach. Part II] [Published in Russian]. Bulletin of surgical gastroenterology. 2009c; 4: 70-77.
5. Bray F, Ren JS, Masuyer E, Ferlay J. Estimates of global cancer prevalence for 27 sites in the adult population in 2012. Int J Cancer. 2013; 132(5): 1133-1145.
6. Caletti G, Ferrari A, Brocchi E, Barbara L. Accuracy of endoscopic ultrasonography in the diagnosis and staging of gastric cancer and lymphoma. Surgery. 1993; 113: 14-27.
7. Caletti G, Fusaroli P. Endoscopic ultrasonography (EUS) and EUS-fine-needle procedures: fascinating technique but little clinical impact. Endoscopy. 2001; 33(6): 537-540.
8. Gono K, Obi T, Yamaguchi M, Ohyama N, Machida H., et al. Appearance of enhanced tissue features in narrow-band endoscopic imaging. J Biomed Opt. 2004; 9: 568-577.
9. Gono K, Yamazaki K, Doguchi N, Nonami T, Obi T., et al. Endoscopic Observation of Tissue by Narrowband Illumination. Opt Rev. 2003; 10: 211-215.
10. Kaprin AD. [Gastric cancer screening: modern opportunities and perspective] [Published in Russian]. Modern achievements in socially significant diseases treatment. 2015. 125-132. URL: http://federalbook.ru/files/FSZ/ soderghanie/Z_16/Z16_2015-Kaprin.pdf
11. Kitabatake S, Niwa Y, Miyahara R, Ohashi A, Matsuura T., et al. Confocal endomicroscopy for the diagnosis of gastric cancer in vivo. Endoscopy. 2006; 38: 1110-1114.
12. Kuvaev RO, Kashin SV, Nikonov EL, Itoh T, Gtoda T. [Early gastric cancer: modern methods of screening, endoscopic diagnosis and minimally invasive treatment] [Published in Russian]. Evidence-based gastroenterology. 2014; 2: 44-51.
13. Loshchenov VB, Linkov KG, Savelieva TA, Loshchenov MV, Model SC., et al. [Hardware and instrumental supply of fluorescent diagnostics and photodynamic therapy] [Published in Russian]. Photodynamic therapy and photodiagnosis. 2013; 3: 17-25.
14. Osawa H, Yamamoto H. Present and future status of flexible spectral imaging color enhancement and blue laser imaging technology. Dig Endosc. 2014; 1: 105-115.
15. Poddubny BK, Kuvshinov YuP, Malikhova OA. [Significance of chromoscopy, endosonography and enlarging endoscopy in solving diagnostic problems precancer and early gastrointestinal cancer diagnosis] [Published in Russian]. Consilium medicum. Modern oncology. 2005; 3: 104-111.
16. Ryabov AB, Sokolov VV, Khomyakov VM, Pirogov SS, Kolobaev IV, Cheremisov VV. [Current approaches to diagnosis and treatment of early gastric cancer] [Published in Russian]. Oncology. PA Gerzen’s Journal. 2015; 2: 71-75. 17. Sokolov VV. [Atlas of videoendoscopy endoluminal surgery in clinical oncology] [Published in Russian]. Moscow. 2015. 152.
18. Testoni PA, Mangiavillano B. Optical coherence tomography in detection of dysplasia and cancer of the gastrointestinal tract and biliopancreatic ductal system. world J Gastroenterol. 2008; 14(42): 6444-6452.
19. Wood WC, Staley CA, Skandalakis JE. Anatomic basis of tumor surgery. Springer-Verlag Berlin Heidelberg, 2010: 265-336.






